 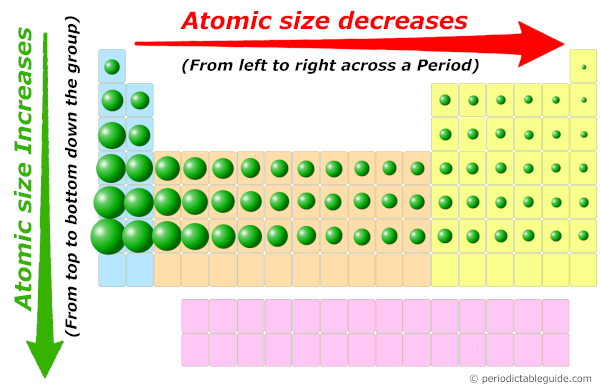
Which particle has the larger radius in each atom/ion pair? Circle your answer. WS 2 Directions: Please answer each fill in the blank with the best answer. Explain the relationship between the relative size of an ion to its neutral atom and the charge on the ions. Name: Date: Unit: Periodic Trends Class period: Atomic Size Trend - WS 2. What is the difference between a cation and an anion? 8. Vocabulary: atomic radius, electron affinity, electron cloud, energy level, group, ion, ionization energy, metal, nonmetal, nucleus, period, periodic trends, picometer, valence electron Prior Knowledge Questions (Do these BEFORE using the Gizmo. Arrange the following in order of increasing atomic radius atomic radius. Why do atoms get larger as you move down a group? 5. The atomic radius is the radius of an atom that is measured from the nucleus to the outermost cloud of electrons.2 Group Period Atomic Radius Trend Atomic radius tends to increase as we go down the periodic table Atomic radius tends to decrease as we go across a period. Element text B B has more electron-occupied shells than element text A A. 
Why do atoms get smaller as you move left to right in a period? 4. Google Classroom Elements text A A & text B B belong to the same group. Explain why you see this trend as you move across a period. Does the same trend hold true Is this the trend you predicted Yes. Choose a couple more periods (excluding 1, 6, and 7). There are some outliers (not addressed in this worksheet). What is the periodic trend for atomic size from top to bottom in a group? From left to right in a period? 3. It trends downward so the atomic radius gets smaller across a period. a) Li or K b) Ca or Ni c) Ga or B d) O or C e) Cl or Br f) Be or Ba g) Si or S h) Fe or Au 2. Which atom in each pair has the larger atomic radius? Circle your answer. below are Periodic Trends Worksheet with Answers, use the periodic table. Download Periodic Trends Worksheet and more Chemistry Lecture notes in PDF only on Docsity!Chemistry ICP for Jan 22: Periodic Trends Use the periodic table, charts, and your knowledge of periodic trends to answer the following questions. As you go down the periodic table, the atomic size trend increasesas you are. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
